Back to Thermodynamics & KTG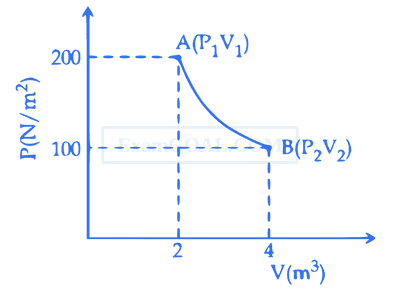
Class 11 • Physics • Chapter-11
Thermodynamics & KTG
Question 552 of 1000
552EasyInteger TypeJEE Mains2021
One mole of an ideal gas at 27$^\circ is taken from A to B as shown in the given PV indicator diagram. The work done by the system will be _________ \times 10 -$1 J. [Given : R = 8.3 J/mole K, ln2 = 0.6931] (Round off to the nearest integer)

Integer / Numeric Answer Type
Enter your answer as a number (integer or decimal).